BVP is pleased to share that portfolio company Corporis Medical has commenced patient enrolment in the clinical investigation of Laprixa, following approval from the Medical Ethics Review Committee (METC) and the Maastricht UMC+ institutional board.
About Corporis Medical and Laprixa
Founded in 2015 and based at the Brightlands Maastricht Health Campus, Corporis Medical is a medical device company specialising in the development and commercialisation of innovative medical devices, delivering the complete product cycle from prototype to clinical studies.
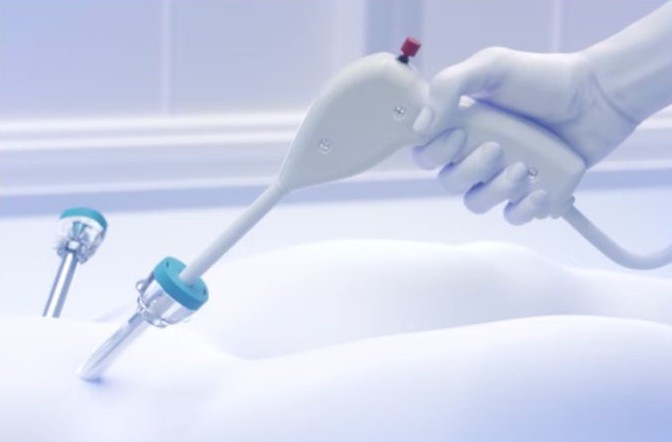
Laprixa is the company’s lead product in surgical wound healing. The Laprixa System is the world’s smallest tissue blood pressure measurement device, enabling objective, quantitative and patient-specific assessment of tissue wound healing capacity. During minimally invasive surgery, surgeons can use Laprixa intraoperatively to determine whether tissue has sufficient blood flow to heal — providing patient-specific insights in real time to help prevent serious complications such as anastomotic leakage.
A significant clinical step
The start of patient enrolment marks the transition from pre-clinical into structured clinical evaluation. With a strong pre-clinical programme already completed, the investigation is designed to assess Laprixa’s real-world clinical value in a controlled, ethics-approved setting at Maastricht UMC+, one of the Netherlands’ leading academic medical centres.
The investigation is being conducted in close collaboration with manufacturing partners Medanco BV and AIM B.V., who have supported device and protocol readiness throughout this stage.
The investigation is supported by the OP-Zuid grant programme, co-financed by the European Union.